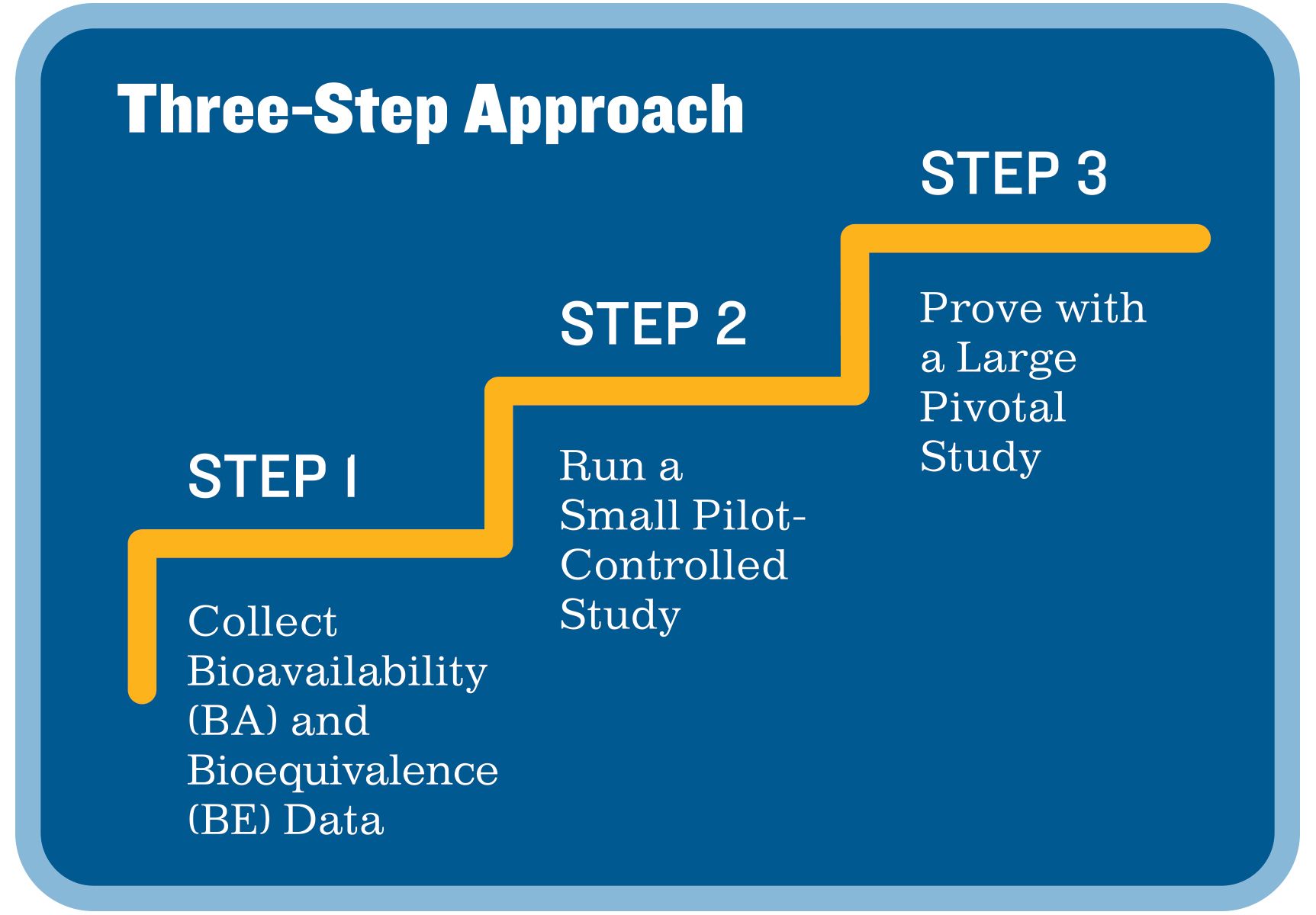
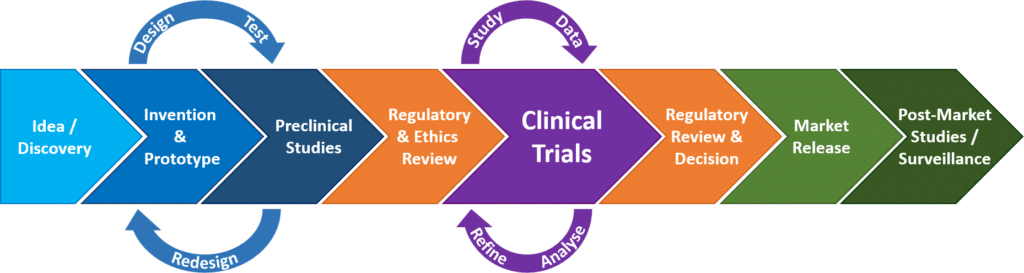
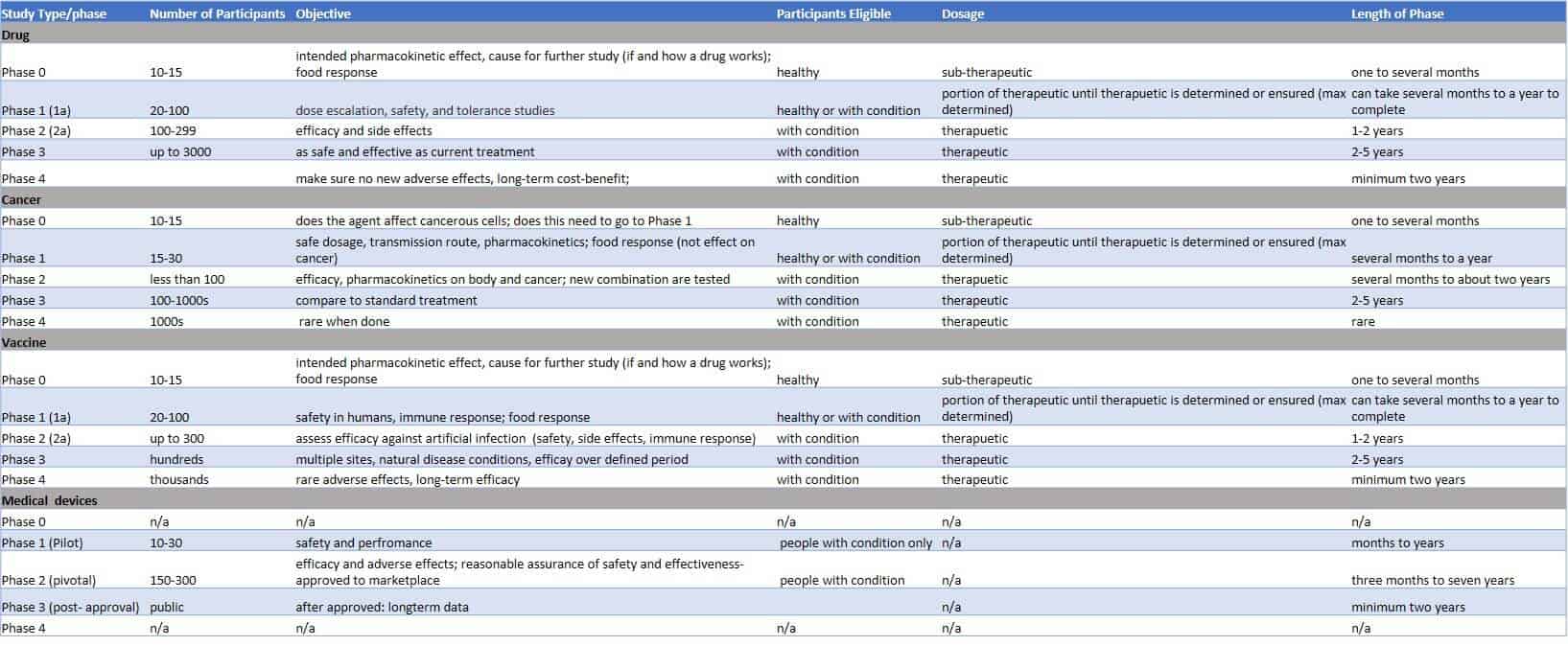
Pivotal Clinical Study to Evaluate the Safety and Effectiveness of the MANTA Percutaneous Vascular Closure Device | Circulation: Cardiovascular Interventions
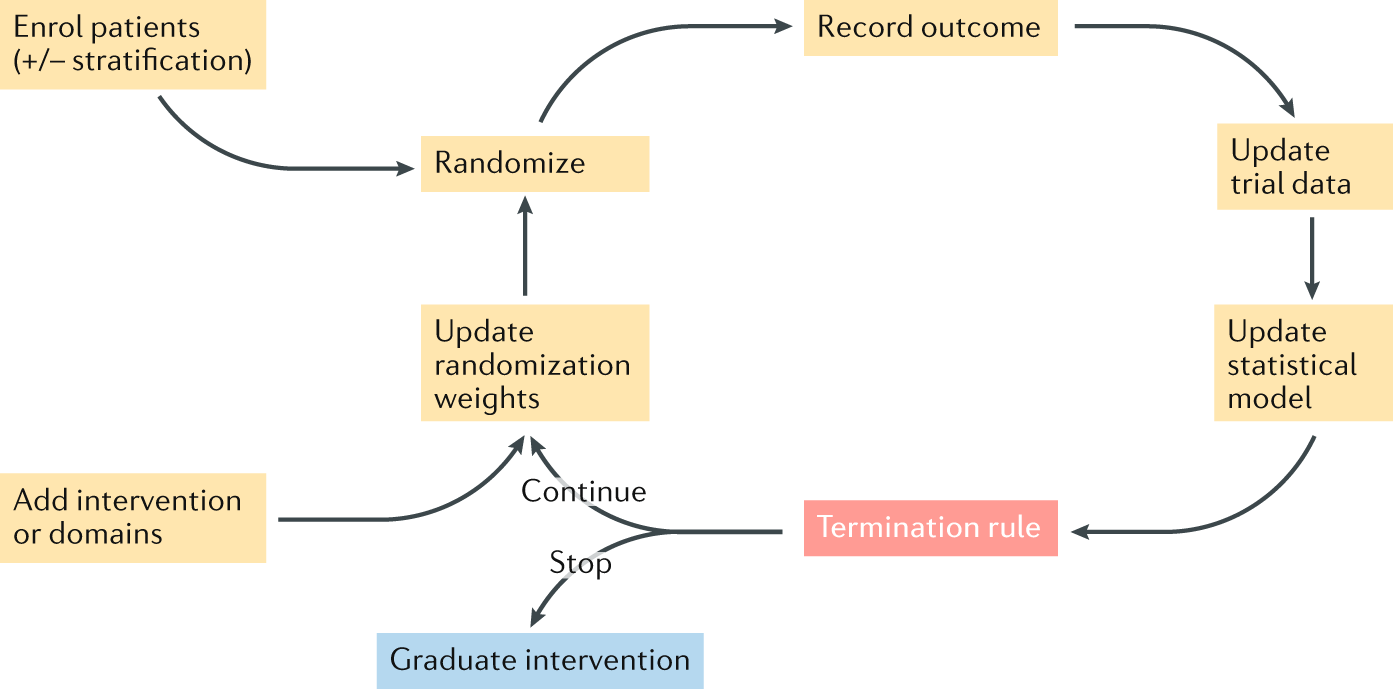
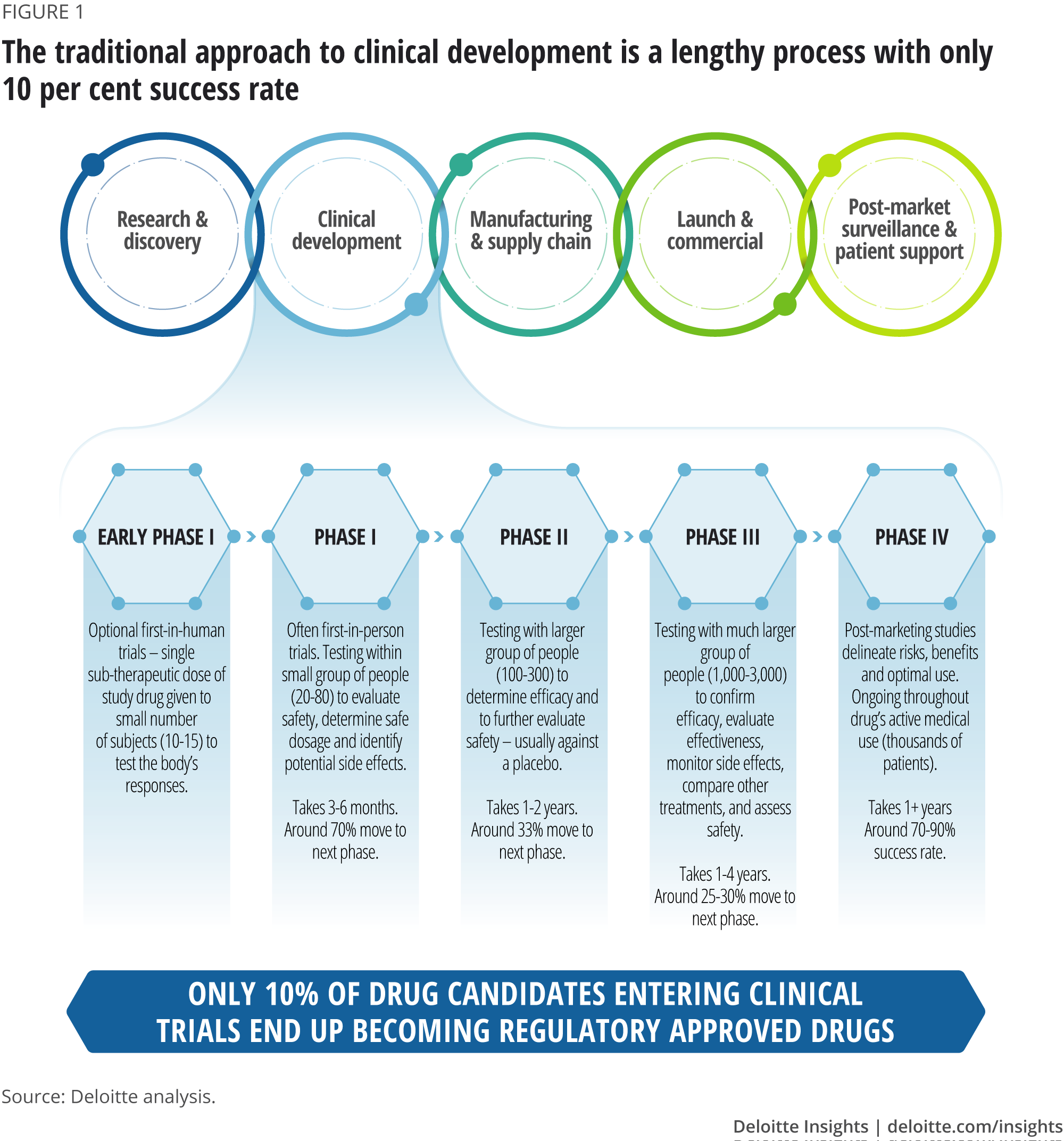
Adaptive platform trials: definition, design, conduct and reporting considerations | Nature Reviews Drug Discovery
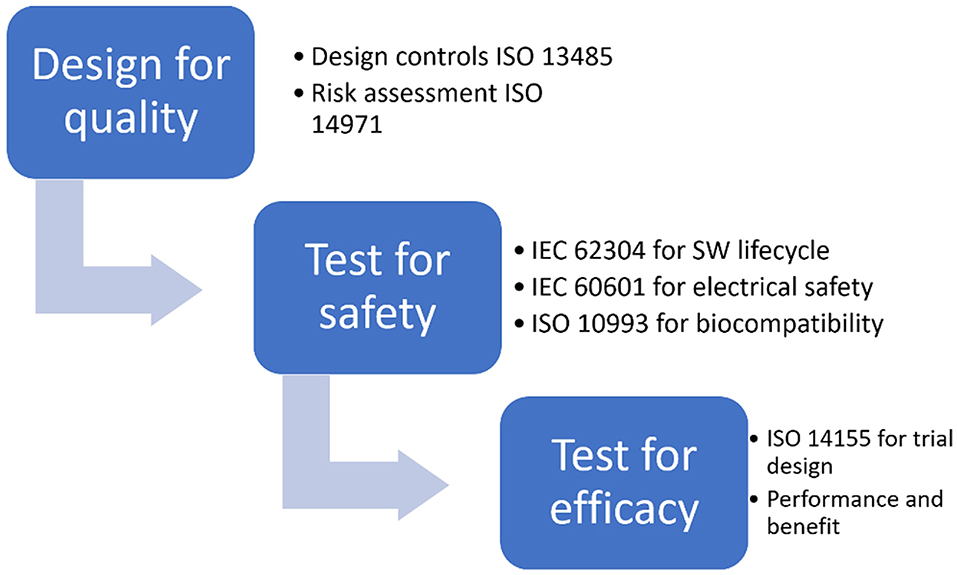
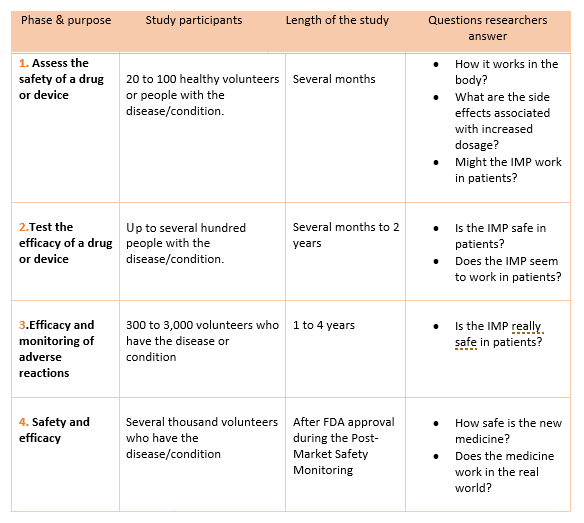
Frontiers | Comprehensive Review on Current and Future Regulatory Requirements on Wearable Sensors in Preclinical and Clinical Testing | Bioengineering and Biotechnology